Our Project – A Vision for Albertan Sulfur
We are Sulfur Us, and our purpose is simple: to create sulfuric acid using an elemental sulfur burning process. In Alberta, only 35% of diluted bitumen produced from oil sands crude is upgraded to synthetic crude oil. This upgrading process sweetens the oil, with the recovered sulfur often being sold as is. We utilize this sulfur, a relatively untapped resource, to create a wide array of sulfuric acid concentrations for various purposes. Such uses include phosphate-based fertilizers, metallurgy, paper pulp plants, and the synthesis of other useful products such as dyes and detergents.
With the various uses of sulfuric acid, its demand has been on the rise, especially for its role in creating fertilizers. We plan to capitalize on this large market to sell our product to various customers, including large markets such as the United States and China. Compounding this, our plant will spur growth in the Albertan economy by encouraging further upgrading of crude oil and opening the door for sulfuric acid production in the province.
Meet our Team!

Morgan McClure
“Our purpose is simple.”

Kurtis Marshall
“Adsorption is a scam.”

Bruce Li
“The numbers look good!”

Kajal Pandya
“Reactors and catalysts and heat, oh my!”
Design Details
PROCESS OVERVIEW
Air is drawn from the atmosphere for the combustion of sulfur. It is filtered, dried, and then purified of all nitrogen via an pressure-swing adsorption process. The sulfur feed arrives to the plant in a liquid form, where it is atomized into a furnace to be reacted with the oxygen to produce sulfur dioxide. Argon leftover from the air adsorption process travels with the sulfur dioxide as an inert gas.
After combustion, the stream proceeds to a double contact catalytic converter (i.e. a fixed-bed reactor) loaded with four beds of vanadium oxide catalyst. The gas makes four passes through this bed to convert the sulfur dioxide into sulfur trioxide. After the third pass, the stream is sent on a detour through the intermediate absorption tower, where the sulfur trioxide reacts with sulfuric acid to produce oleum, which is used later to produce more sulfuric acid. The sulfur trioxide stream then returns to the reactor for its fourth and final pass; a lower concentration of sulfur trioxide from its trip through the intermediate absorption tower means a higher conversion for its final pass, as the reaction is pushed “to the right”.
The stream then enters a fluidized bed reactor, utilizing more vanadium oxide catalyst, that pushes the conversion to virtually 100%. It then goes to the final absorption tower, which creates oleum in the same manner as the intermediate absorption tower. This oleum, or “fuming sulfuric acid”, is taken to an acid pump tank, where it is diluted into the standard high purity (~98% sulfuric acid). Some of this acid is sent across the plant for unit operations (absorption towers and for air drying), while the rest is sent to be diluted and stored for sale.
Catalytic Converter Specifications
The catalytic converters are one of the most significant parts of the process. Our design has improved the oxidation of the sulfur dioxide with low pressure drops, by using the new vanadium oxide catalyst. Intalox ceramic saddles packing also assist in providing high surface area for the reaction and reduce pressure drops. With controlled temperatures of the reactor, the catalyst activity will be maximized. Our design has two reactors, a fixed bed reactor with the overall conversion of 95.90%, and a fluidized bed reactor to push the conversion to virtually 100%.
The oxidation of sulfur dioxide is a highly exothermic reaction and hence it is vital to control the operating temperatures for the catalyst’s maximum activity. Our design solution is to use Waste Heat Boilers for inter-bed cooling and produce high quality energy, high pressure steam. With inter-bed cooling, the catalyst will push to the maximum conversions at operating temperatures.
The material of construction for these converters will be robust and reliable. Material selection was finalized to be SS-304H based on the acceptable stress values at high temperatures and corrosion resistance to the fluid. Using insulation and process control systems, the operating conditions will be controlled to maximize the conversion in the reactors.
HOW OUR DESIGN ADDRESSES PRACTICAL ISSUES
Did you know that Alberta has stockpiles of sulfur so large that you can see them from Google Maps?
A source of this sulfur is the upgrading of Albertan diluted bitumen, where the sulfur is recovered during the sweetening process as raw hydrogen sulfide gas before being converted to its elemental state. Only about 35% of this bitumen is upgraded. Sulfur Us finds a more productive and economic use for this sulfur by purchasing it and converting it into sulfuric acid, incentivising further bitumen upgrading and creating a new, valuable export for the province.
Sulfuric acid is a very valuable export. The global production of it was ~284 million metric tonnes as of 2019, which is expected to increase by 1.5% by 2025. With the ever-increasing need for food around the world, it should be no surprise fertilizer production is the primary use of the acid, accounting for ~57% of its market share as of 2017. The acid produced consequently has a very practical and needed purpose in the real world, all while being produced well below environmental standards.
WHAT MAKES OUR DESIGN INNOVATIVE
NOx emissions are one of the main pollutants sulfuric acid plants produced. Our design, by using our oxygen-generating pressure swing adsorption unit, removes nearly all nitrogen from process air prior to combustion. This virtually eliminates NOx pollutants from our plant, an innovation which makes our process that much greener.
The source of our feedstock has been largely untapped in Alberta and would serve to invigorate a new sulfuric acid industry for the province. A welcome bonus to creating said industry would be increased incentive to upgrade more Albertan bitumen, which stems from increased purchase of sulfur from these upgrade facilities.
The double contact process that we use in our plant ensures that virtually 100% of the sulfur dioxide is converted to sulfur trioxide, maximizing product yield.
WHAT MAKES OUR DESIGN SOLUTION EFFECTIVE
- The sulfur we use for our feedstock would otherwise be sold as-is or simply stockpiled. Our design finds an effective and profitable use for this sulfur.
- Other than the aforementioned removal of virtually any NOx emissions, careful attention has been given to the environmental impacts of the plant to ensure that it is well below Alberta guidelines for pollution.
- We can alter our product’s concentration to fit a wide variety of needs. For example, sulfuric acid with 25% w/w purity is ideal for phosphate-based fertilizer production, which makes up one of the top uses of sulfuric acid.
- The double contact process we use to convert sulfur dioxide to sulfur trioxide operates with virtually 100% conversion.
- The vanadium oxide catalyst has the highest activity at our operating temperatures and thus rarely has to be replaced or regenerated.
HOW WE VALIDATED OUR DESIGN SOLUTION
Both hand calculations and simulation software was used to validate our design. This includes material and energy balances, unit sizing, and thermodynamic/equilibrium calculations. The simulation software, Symmetry, was given data generated by our hand calculations to give an estimation of the results of each process units. Our values were also compared to literature and industry standards.
FEASIBILITY OF OUR DESIGN SOLUTION
Our design was created using several key assumptions to simplify and streamline the calculations. These include ideal gas law, Langmuir adsorption, complete combustion, etc. This limits the ability of the design to be applied to a real-life scenario; running experiments for empirical data, erecting a small-scale plant, and consulting a wide array of industry experts would increase its feasibility greatly.
From an economic point of view, our design is incredibly feasible, with a payback period of ~42 months and a cash flow of ~282 million CAD after a 10-year operation life.
Partners and Mentors
A very special thanks to our supervisor, Dr. Maen Husein, who always kept us on our toes and heading down the right track throughout the year. Another big thank you goes out to our professor, Dr. Hector De la Hoz Siegler, whose hard work and perseverance made these courses a reality.
Gallery
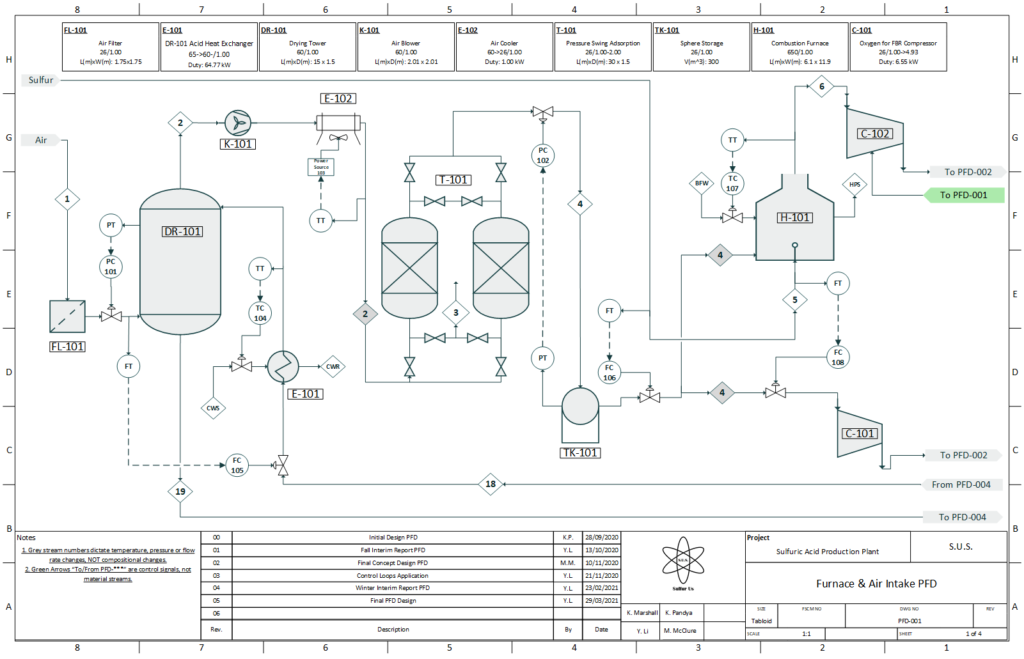
PFD 1 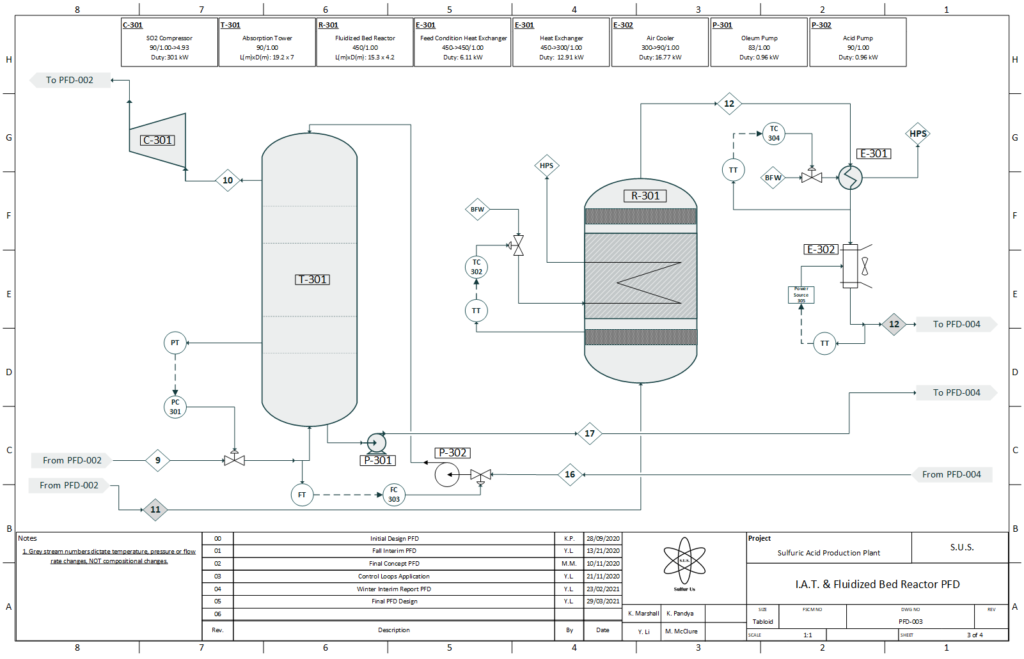
PFD 3 
PFD 4 
PFD 2 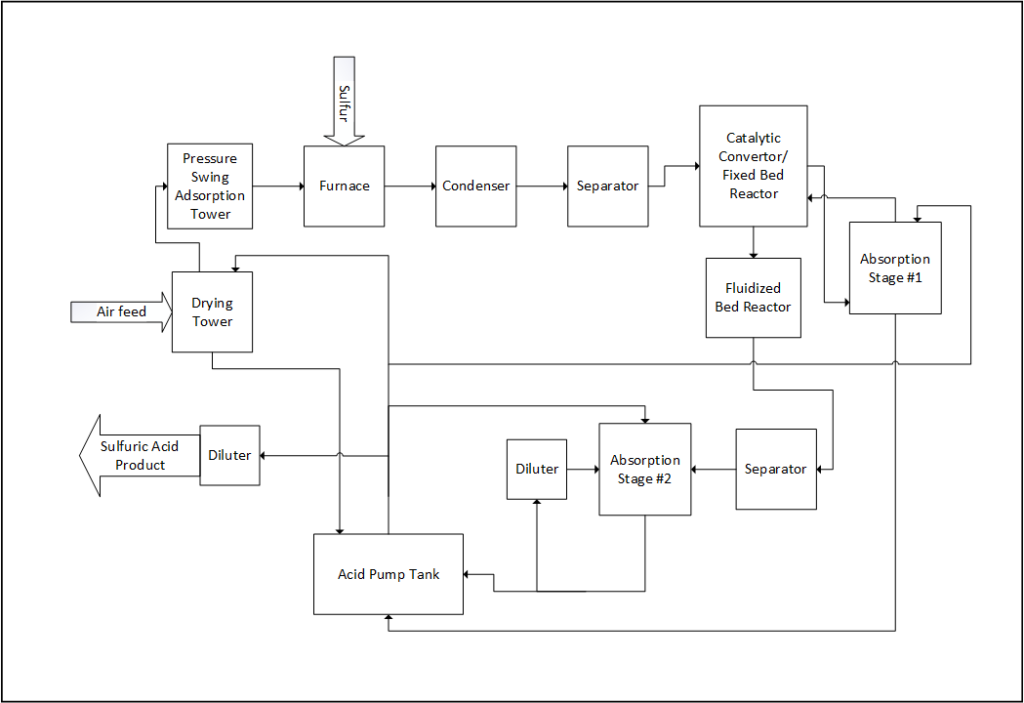
BFD