Project Category: Multidisciplinary
Join our presentation
About our project
The BioNorth Solutions (BNS) company is a relatively new bioremediation company that specializes in plant growth and hydrocarbon clean-up products with the use of microbes. They have four microbe products called PGP4, PGP2R, Microbiate SG and Microbiate HS. BNS has provided a cost effective solution to oil spill clean-up and work-site rehabilitation by spreading and adding Microbiate to the soil (SG) or hard surface (HS). When the Microbiate formula is added to the soil the microbes are able to ‘eat’ and eliminate the hydrocarbons. The next products are PGP4 and PGP2R, they are used to increase plant growth for large and small scale farmers. After testing the PGP products, it was found that it is able to increase the yield of farmed crops by 30%. With projected sales to be in the millions in the next 3 years, the company finds themselves at a crossroads when it comes to their future of manufacturing. There are two options:
- To continuously pay a 3rd party manufacturer to create, assemble and package their product with hopes of their infrastructure meeting future demand
or
- To construct their own flagship manufacturing plant that will be capable of meeting all demand, however this may cost a large portion of the company’s capital cost
Our capstone engineering team comes in to design and provide a cost analysis of the main process in the manufacturing plant. Our work will be submitted to the BNS team to see when an internal flagship manufacturing plant is viable to construct.

About our sponsor (BioNorth Solutions Inc.)
BioNorth Solutions Inc. is a Federally registered, Canadian manufacturing company, specializing in microbial science, more specifically, the development and production of “packaged microbes”. Their proprietary microbes provide hydrocarbon soil remediation and plant growth enhancement solutions worldwide.
Meet our team members
Details about our design
HOW OUR DESIGN ADDRESSES PRACTICAL ISSUES
In this process, there were a few main points that were practical issues that needed to be addressed. The following points are what the project was revolved around and fully solved throughout the development of the plan:
- There needed to be enough production capacity to handle expected growth
- The entire plant is to be automated and functioning independently of any major manual labour
- Any sort of negative affect on the quality of the product is to be minimized completely, especially any sort of cross-contamination
The entirety of the process was split into smaller stages to be able to further work on the automation of the entire process from beginning to end. Each stage was then consulted with experts, such as vendors from companies specializing in bioreactors, mixing, drying and packaging, to ensure that each machine has certain capabilities that allow for desired production and minimal manual labour. The bioreactors for example have clean-in-place systems that allow for extended run times of the process, the mixers are able to provide minimal down-time as well and with the interconnection between all the machines using either piping or conveyors that are then able to lead to the dryers and from there to packaging, the product is only manually worked on towards the end of the process to then proceed to distribution.
The characteristics of the chosen equipment was also able to address all concerns for cross-contamination and any risk to the integrity of the end product as at any point during operation hours, majority of the equipment is able to be self-cleaned within a small period of time to ensure that major revenue loss can be avoided. Another important approach that was exercised during the development of this facility was the careful organization of the process that allowed for all products to never come in contact with one another allowing for complete isolation and preservation of the Microbiate and PGP products.
WHAT MAKES OUR DESIGN INNOVATIVE
Our capstone introduced us to four new innovative products: hydrocarbon remediators, hard surface adsorbents and two plant growth promoters. Our project’s very nature is built on new products that require an innovative approach to creating a large-scale manufacturing process design with the CEO, engineers, bioscientists, and company investors. The products increase plant growth by 30%, increase resistance to bacterial and fungal pathogens and reduce fertilizer use by 20%. Our products and process design are vital to meeting the increased 70% food production for the projected 9.1 billion people by 2050. Two of our products can operate from 0.1 C to 49 C and save 30% to 50% in cost instead of using the dig and dump method in the oil & gas industry while adding hydrogen, oxygen, and only trace amounts of CO2 back into the environment. And, we are just getting started on the impact and innovation of our project!
Our design’s bioreactor stage includes the ability to thoroughly flush out contents inside the vessel if a sign of impurity and contamination is detected further downstream, which allows minimizing wastage of product, money and meeting a high standard of a quality product. Another innovative aspect of our microbe growth method is the two-stage system. In stage 1 of microbe growth, the bacteria is bulk grown. The second stage is specialized to nourish the grown bacteria ensuring the effectiveness of our bacteria and product.
Our sensitive bacteria also needed extra care in the two-stage mixing process. The first stage uses a smaller container to form a well-mixed bacteria concentrate. Using this unique step, we were able to mix a bioproduct with the more resistant and abrasive agricultural products at a microscopic level. (Reference to the pictures below to understand the magnitude of ingredients we are dealing with).

We overcame a huge challenge with our innovative design, finding a dryer that reduces the mixer outlet product’s water content from 50% to about 10% while maintaining the temperature consistently below 50 °C for our temperature-sensitive bacteria. We chose a rotary dryer due to its ability to break up the wet pastelike material with its agitation and an inlet air temperature at 200 °C. We selected a continuous conveyor dryer for the other product because of its ability to grow modularly and accurate temperature control.
Our process’s innovation doesn’t stop there. Our bioproduct manufacturing process required automation to mitigate contamination in the facility while meeting our products’ great demands. We used a flex packaging machine to package our products in different bag sizes (18kg – 55kg) at a speed of up to 6 bags per minute and a container packaging machine at up to 60 containers per minute to cater to end-users and business to business customers. Our modular packaging station is automated from bag alignment, filling, sealing and palletizing. We also have a simple automated switch to change all our products’ labelling from BNS label to white label packaging.
We have covered the innovation and creativity in each of our signature design steps. While dealing with material of various sizes, we showed creativity by choosing less than an inch size pipe for our all wet biomaterials and choosing enclosed customized conveyor belts to transfer dry material with sludge and powder-like consistency. It is incredible to see our mixers have both piping and closed conveyor connections.
WHAT MAKES OUR DESIGN SOLUTION EFFECTIVE
The BioNorth Solutions flagship manufacturing plant is a state-of-the-art design that features modular construction with fully automated production lines. Each piece of equipment has been carefully specified to production forecasts with opportunities for growth.
The four production lines work simultaneously to produce and package four distinct products with minimal downtime. A detailed plant layout allows for optimal flow of product, while flow rates have been calculated to produce zero excess.
The ability for our design to pivot and expand as consumer demand changes highlights the effectiveness and lasting attributes of this process design.
HOW WE VALIDATED OUR DESIGN SOLUTION
For this design we were provided Pro Forma data from our sponsor. Our task included designing a manufacturing plant to meet these sales predictions with ability to expand production if needed.
Once the production process was determined, the team calculated mass and volume flow rates for each of production lines. These were validated with the BNS team allowing us to move forward with equipment research and selection.
The team compiled a vendor list of appropriate equipments (bioreactors, mixers, dryers, packagers). Each of these equipments was compared on cost, capability and efficiency. There was thorough communication with equipment vendors to ensure an optimal system was being designed.
All equipment data sheets are readily available to validate production requirements. The manufacturing process is designed in such a way that additional pieces of equipment can be introduced should demand greatly vary from Pro Forma numbers.
FEASIBILITY OF OUR DESIGN SOLUTION
The feasibility of our design boils down to two factors.
- Does it make sense from an economic standpoint.
- Does this process make sense physically. (will the process even work and will it have a reasonable footprint)
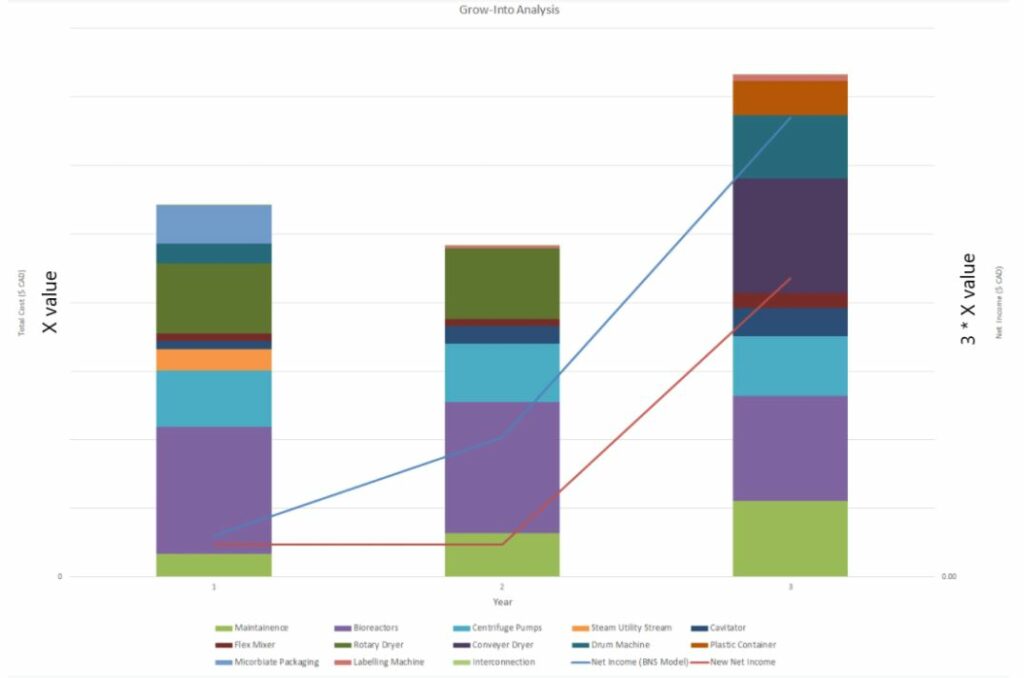
- On the economics side we were able to do a “grow into analysis” which allowed the manufacturing equipment to be purchased over several years as production ramps up.
- This analysis shows provided the chart below. This chart shows us the total costs and expected net income over 3 years.
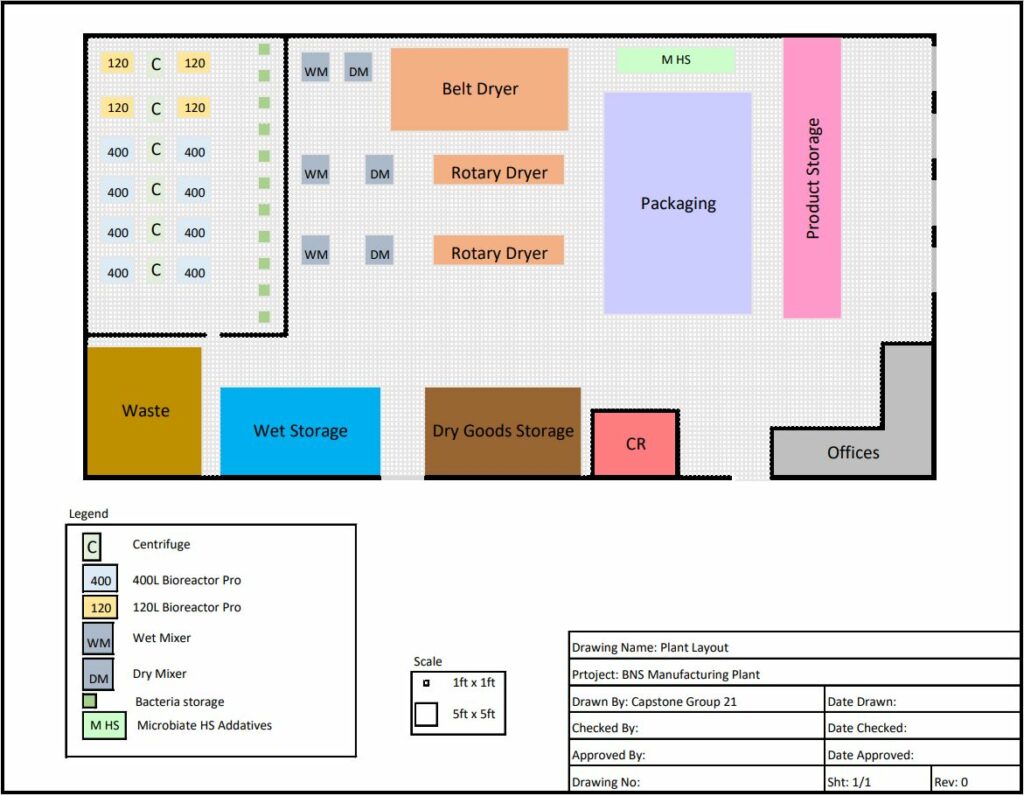
- On the physical side of thing we needed to ensure that all the proposed equipment would not only meet the requirements to meet production but also fit within the proposed 20,000 sq ft facility.
- The data validation shows that the equipment will meet the required demand while the proposed plant layout shows that all the equipment will fit with some room for expansion.
Partners and mentors
We want to thank the many people who helped us with this project.
- We would like to thank Dr. Paul Tu for his technical and project advice.
- We would like to thank Dr. Roes (Arief) Budiman and Jaehyun Yang for project guidance.
- We would like to thank the BioNorth Solutions CEO, Brad Klock, for initiating, sponsoring, and guiding the project.
- We would like to thank the BioNorth Solutions EVP of Sales and Business Development, Dan Bissonette, for supporting and encouraging the project.
- We would like to thank the BioNorth Solutions Biologists and Co-Founders, Dr. Miranda Lock and Amber Kivisto, M.SC, for the project’s scientific support.





